Measuring our greenhouse gases more rapidly and accurately
Our team aims to miniaturise the current bulky gas sensing equipment and use it in remote areas like Australia’s outback.
The challenge:
Gas sensing equipment is bulky, expensive and confined to laboratories
From detecting explosives to measuring the amount of carbon dioxide in our atmosphere, being able to sense gases accurately has enormous ramifications across environmental and defence industries.
The current instruments that detect gases are extremely effective at sensing even tiny samples – even one part in a billion – of any given gas. This is thanks to the large library of chemicals being well documented already – where we have a reliable encyclopaedia of ‘chemical fingerprints’ to measure against.
However, while this gas detection equipment is very expensive and sensitive, relatively slow and so bulky that it remains confined to complex laboratories, where only specialists know how to operate them. This limits the potential of these tools, rendering them too bulky to be used in airports for national security, and too large to be deployed when measuring gases for long range field experiments.
Our response:
Using the world’s most accurate ruler for light to make gas sensing more rapid
The gas sensing equipment achieving these very accurate results in the laboratory is underpinned by an approach called absorption spectroscopy.
This technique works by shining light through a gas sample, and if the gas in question is present, then it will hit its specific chemical signature (or colour) and absorb that colour. For example, the chemical signature of methane (CH₄), carbon dioxide (CO2) and water (H₂O) are all unique, as seen in the table on the right.
In the past, this technique relied on only one gas being measured at a time – where you would include the testing gas and a control gas as a point of reference. Then by looking at the contrast between the two colours, you could identify which gas was in the sample. However, this approach was relatively slow and inefficient.
To gather clues about the kind of gas in your sample, you would also need to tune your light source so it would ‘compute’ with the type of gas you were looking at. For example, to detect carbon dioxide, you would need to make sure you used a 4.3 µm mid-infrared laser, but for an explosive gas like methane, you would need to use a 3.5 µm laser instead.
However, the new method of combining two techniques – called absorption spectroscopy and the optical frequency microcomb – allows for many gases to be measured at the same time.
This is because the optical frequency comb includes all the potential colours and gases you could possibly test, and separates them out into equally spaced colours, like the teeth on a comb. It also has such a broad reference base that it can automatically correct for the type of light needed for a specific gas, simply by scanning the laser.
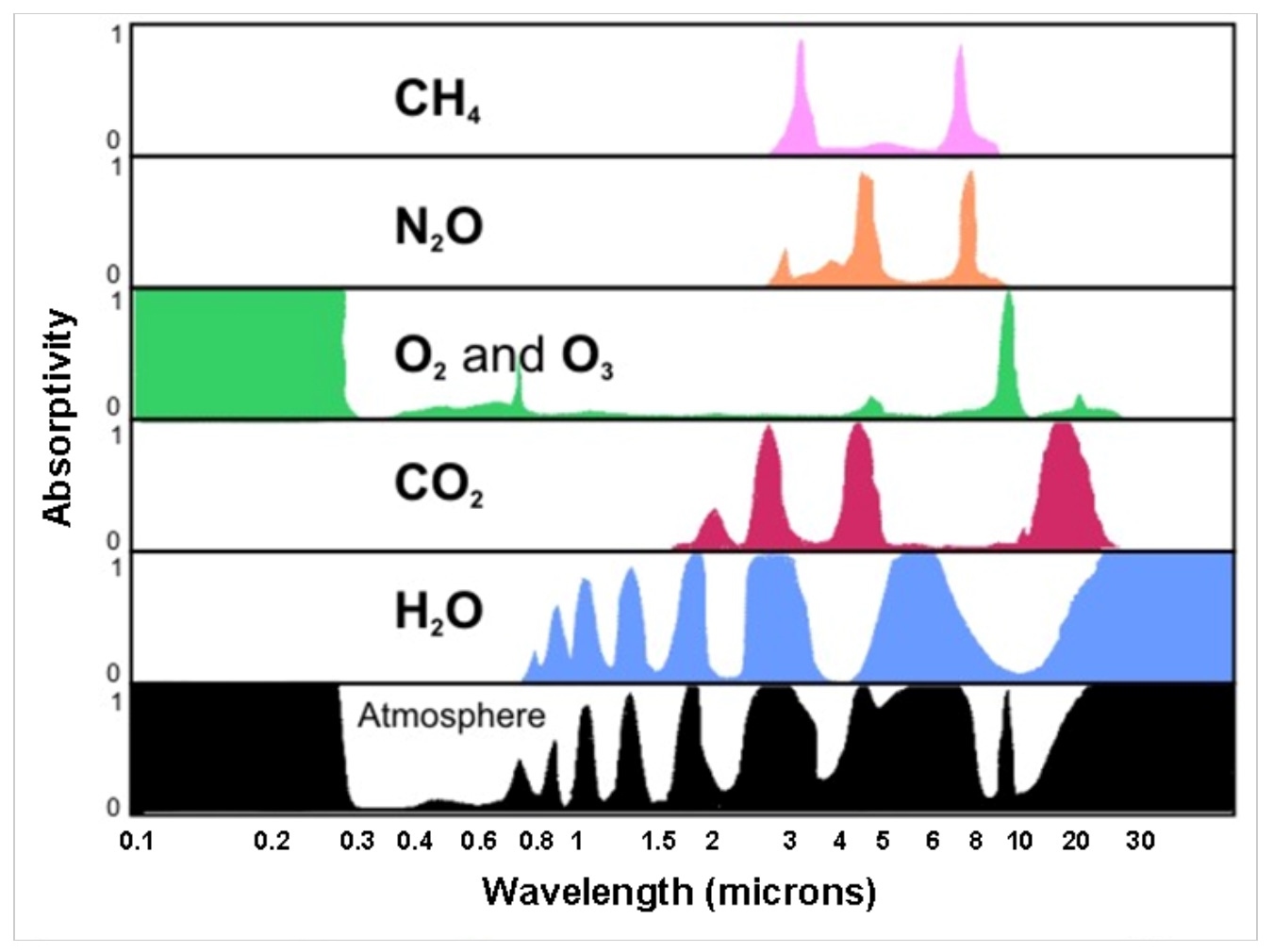
As light hits different gas samples, a different chemical signature (or colour) is absorbed. In this image you can see the unique chemical makeup of methane (CH₄), nitrous oxide (N₂O), oxygen (O₂) and ozone (O₃), carbon dioxide (CO₂) and water (H₂O) – and how these gases make up our atmosphere. Image credit: David Babb.
The expected results:
Our team at the Centre in Optical Microcombs for Breakthrough Science (COMBS) plans to develop a portable and small device with less moving parts, that retains the sophisticated techniques of the bulky equipment in the lab.
We plan to create these frequency comb devices on a chip that are only a few centimetres in size, to be packaged in a box to make it an isolated, stable and reliable gas sensor in any environment – from airports for explosives detection to the outback Australia for environmental sensing.
The team are developing ways to create complex but informative patterns of light in scattering media – this scrambled pattern of light is incredibly sensitive to changes in the gas. This combined with the microcomb technology could herald new compact gas sensing devices.
With the interdisciplinary expertise spanning designers, fabrication specialists, and customers from defence and environmental industries, we have a full pipeline of experts to understand the intricacies of the entire industry.

The teams' expected results are to create portable gas sensing equipment for airports and field experiments.


